PHARMA NEWS BD, Special Manufacturing Report.
Dhaka, Bangladesh – In a major step toward strengthening pharmaceutical manufacturing knowledge and compliance awareness, a comprehensive technical and regulatory summary has been developed covering the complete lifecycle of Rotary Tablet Compression Machines, including engineering science, GMP validation, risk management, inspection readiness, and full documentation systems.
This initiative is designed to support pharmaceutical manufacturers, QA professionals, validation teams, and regulatory bodies in ensuring safe, efficient, and compliant solid oral dosage production.
Complete Tablet Manufacturing Workflow
The documentation outlines the full manufacturing process from raw material receipt to finished product release:
-
Raw Material Control
-
API and excipient receipt, quarantine, QC testing, and release.
-
Compliance with WHO GMP and quality verification standards.
-
-
Dispensing & Weighing
-
Controlled dispensing areas with dust extraction.
-
Double verification and batch record documentation.
-
-
Granulation Process
-
Wet granulation (RMG + FBD)
-
Dry granulation (Roller Compactor)
-
Direct compression methods
-
Optimization for flowability and compressibility.
-
-
Final Blending
-
Controlled lubricant blending.
-
Prevention of over-lubrication to maintain hardness integrity.
-
-
Tablet Compression
-
Die filling, pre-compression, main compression, and ejection.
-
In-process monitoring every 30 minutes (weight, hardness, thickness, friability).
-
-
Dedusting & Metal Detection
-
Removal of residual powder.
-
Metal detection with auto-rejection system.
-
-
Coating & Packaging
-
Film coating systems.
-
Blister/bottle packaging with controlled storage.
-
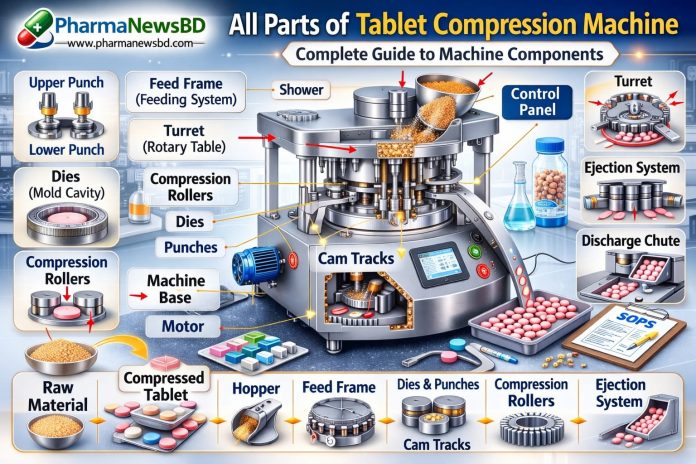
Rotary Tablet Compression Machine – Parts & Functions
The rotary tablet press consists of critical components, each serving a defined mechanical function:
-
Hopper – Stores granules for compression.
-
Feeder System – Ensures uniform die filling.
-
Die – Defines tablet shape and volume.
-
Upper & Lower Punches – Apply compression force.
-
Pre-Compression Roller – Removes air entrapment.
-
Main Compression Roller – Forms tablet under high pressure.
-
Turret – Rotates tooling stations.
-
Ejection Cam System – Safely ejects finished tablet.
-
Load Cells & PLC/HMI – Monitor compression force and process parameters.
Advanced Compression Engineering Science
The documentation includes material science fundamentals:
Tablet Formation Mechanisms
-
Plastic deformation
-
Elastic recovery
-
Fragmentation bonding
Key Scientific Models
-
Heckel Equation – Powder densification analysis.
-
Kawakita Equation – Volume reduction modeling.
-
Dwell time influence on tensile strength.
Process Optimization Parameters
-
Compression force
-
Turret speed
-
Pre-compression ratio
-
Lubricant percentage
-
Environmental humidity (45–55% RH recommended)
Risk Assessment & FMEA Framework
A structured Failure Mode and Effects Analysis (FMEA) was developed for:
-
Weight variation (RPN evaluation)
-
Capping and lamination
-
Cross-contamination risks
-
Tooling wear
-
Safety interlock failures
Risk mitigation strategies include:
-
Automatic weight monitoring
-
Cleaning validation
-
Preventive maintenance programs
-
Calibration control systems
WHO GMP Inspection Readiness
A complete inspection guide was prepared covering:
-
Equipment qualification (IQ/OQ/PQ)
-
Cleaning validation and MACO limits
-
In-process control documentation
-
Operator training records
-
Change control procedures
-
CAPA and deviation management
The framework ensures readiness for inspections by:
-
WHO GMP
-
US FDA
-
EU GMP
-
DGDA Bangladesh
Validation Master Plan (VMP) Structure
The validation system includes:
-
Design Qualification (DQ)
-
Installation Qualification (IQ)
-
Operational Qualification (OQ)
-
Performance Qualification (PQ)
-
Cleaning Validation
-
Revalidation Triggers
Statistical evaluation methods such as Cp/Cpk analysis are incorporated for process capability assurance.
Complete GMP Documentation Bundle
A fully structured digital documentation package was developed, including:
-
SOPs for operation, cleaning, maintenance, calibration
-
IQ/OQ/PQ templates
-
Cleaning validation protocols
-
GMP audit checklists
-
FMEA risk assessment sheets
-
Deviation & CAPA templates
-
Batch Manufacturing Record (BMR) templates
-
In-Process Control logs
-
Statistical QC tools
-
Operator training manuals
This bundle is audit-ready, editable, and deployable for pharmaceutical facilities seeking regulatory compliance and operational excellence.
Commitment to Pharmaceutical Excellence
This comprehensive technical and compliance framework reflects a strong commitment to:
-
Manufacturing precision
-
Patient safety
-
Regulatory compliance
-
Engineering excellence
-
Continuous quality improvement
By integrating advanced compaction science with GMP documentation and inspection preparedness, the initiative strengthens solid dosage manufacturing standards across the industry.
Advanced Rotary Tablet Compression Technology Strengthens GMP Compliance
Engineering Excellence, Validation Science, and WHO Inspection Readiness Integrated into One Comprehensive Framework
In a significant advancement for pharmaceutical manufacturing, a comprehensive technical and regulatory framework has been introduced covering the complete lifecycle of Rotary Tablet Compression Machines. The initiative integrates engineering science, Good Manufacturing Practice (GMP) compliance systems, validation master planning, inspection preparedness, and structured documentation to support modern solid oral dosage manufacturing.
The framework aims to strengthen pharmaceutical production quality, regulatory transparency, and operational excellence across manufacturing facilities.
From Raw Material to Finished Tablet: The Complete Manufacturing Workflow
Tablet production begins with strict raw material control. Active Pharmaceutical Ingredients (API) and excipients undergo quarantine, laboratory testing, and QA approval before release. Controlled dispensing areas ensure accurate weighing and documentation in the Batch Manufacturing Record (BMR).
Granulation follows, using either wet granulation (RMG + FBD), dry granulation (roller compaction), or direct compression techniques. Each method optimizes powder flow and compressibility, ensuring uniform tablet formation.
After final blending—carefully managing lubricant ratios to prevent hardness reduction—the material proceeds to the rotary tablet compression stage.
Inside the Rotary Tablet Compression Machine
At the core of the process is the high-precision rotary tablet press. This sophisticated machine converts pharmaceutical powder into accurately dosed tablets through synchronized mechanical movements.
Key Components and Functions:
-
Hopper – Stores and feeds granules into the system
-
Feeder System – Ensures consistent die cavity filling
-
Die – Determines tablet shape and volume
-
Upper & Lower Punches – Apply compression force
-
Pre-Compression Roller – Eliminates air entrapment
-
Main Compression Roller – Produces final tablet hardness
-
Turret – Rotates tooling stations at controlled speed
-
Ejection Cam – Safely releases finished tablets
-
Load Cells & PLC System – Monitor compression force and process parameters in real time
In-process quality control is conducted at 30-minute intervals, monitoring weight, hardness, thickness, and friability to ensure compliance with specifications.
Advanced Compression Engineering Science
The framework incorporates fundamental material science principles to optimize tablet strength and integrity:
Material Behavior During Compression
-
Plastic deformation
-
Elastic recovery
-
Particle fragmentation
Scientific Models Applied
-
Heckel equation for densification analysis
-
Kawakita equation for volume reduction modeling
-
Dwell time analysis for tensile strength optimization
Critical process parameters such as compression force, turret speed, pre-compression ratio, lubricant percentage, and environmental humidity (recommended 45–55% RH) are carefully controlled.
Risk Assessment and Quality Assurance
A structured Failure Mode and Effects Analysis (FMEA) evaluates potential risks including:
-
Weight variation
-
Capping and lamination
-
Cross-contamination
-
Tooling wear
-
Safety interlock failure
Preventive maintenance schedules, cleaning validation protocols, calibration programs, and statistical process control (Cp/Cpk analysis) form part of the comprehensive risk mitigation strategy.
WHO GMP Inspection Readiness
Inspection preparedness is central to the initiative. The compliance framework includes:
-
Installation Qualification (IQ)
-
Operational Qualification (OQ)
-
Performance Qualification (PQ)
-
Cleaning Validation with MACO limits
-
Deviation and CAPA systems
-
Change control procedures
-
Operator training documentation
This ensures readiness for audits by WHO GMP, US FDA, EU GMP, and DGDA Bangladesh authorities.
Complete GMP Documentation Bundle
To support implementation, a fully structured digital documentation package has been developed, including:
-
Standard Operating Procedures (SOPs)
-
Validation Master Plan (VMP)
-
IQ/OQ/PQ templates
-
Cleaning validation protocols
-
FMEA risk assessment sheets
-
Batch Manufacturing Record templates
-
Statistical QC tools
-
Audit checklists
-
Operator training manuals