PharmaNewsBD: Tablet Processing Checklist, designed to support manufacturers in ensuring consistent quality, regulatory compliance, and operational efficiency across all stages of tablet production.
Process validation for the tablet manufacturing process
- Is the reason for the presence of each ingredient in the formula provided?
- Are the normal properties of each ingredient mentioned? Are these properties used or changed in the formula under study.
- Are the characteristics of the initial powder blends, the wet and/or dry granulations and the final blends mentioned?
- Is the density – ‘loose’ and ‘tap’ determined?
- Is the particle size distribution determined?
- Is the surface area determined?
- Are the flow properties, e.g. contact angle determined?
- Are the moisture content, if applicable determined?
- Are all the processing steps needed for the initial scale-up determined?
- Is the optimal blending time-based MFR.
- Are the tests used to assess the uniformity of the final product determined? Content uniformity, weight variation testing
The checklist provides a step-by-step framework covering the full tablet manufacturing workflow, from raw material handling to final packing, in alignment with WHO GMP and Bangladesh DGDA standards.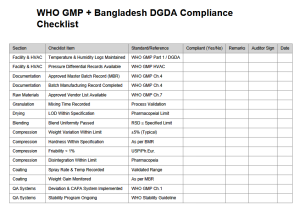
GMP-Based Audit Checklist (Tablet Manufacturing)
A. Facility & Premises
-
HVAC qualification & pressure differentials recorded
-
Temperature & humidity logs maintained
-
Cleanroom classification maintained
-
Pest control records available
-
Proper material & personnel flow
-
Segregation of approved/rejected materials
B. Equipment
-
Equipment IQ/OQ/PQ completed
-
Calibration status within validity
-
Preventive maintenance schedule followed
-
Cleaning validation available
-
Status labels attached (Clean/In Use/Under Maintenance)
C. Documentation
-
Approved Master Batch Record (MBR)
-
Batch Manufacturing Record (BMR) completed
-
Logbooks updated
-
Deviation handling system in place
-
Change control records maintained
-
SOPs current & controlled
D. Raw Materials
-
Approved vendor list available
-
COA verified
-
Quarantine & release system followed
-
Sampling procedure compliant
E. In-Process Controls
-
Blend uniformity performed
-
LOD within limit
-
Weight variation controlled
-
Hardness & friability tested
-
Disintegration & dissolution performed
F. Personnel
-
Training records available
-
GMP training up-to-date
-
Gowning procedure followed
-
Health monitoring records maintained
G. Quality Control
-
Analytical methods validated
-
Stability program running
-
OOS procedure implemented
-
Retention samples stored properly
Pre-Operation
-
Line clearance completed
-
Equipment cleaned & labeled
-
Calibration verified
-
MBR available
Dispensing
-
Materials verified
-
Weights cross-checked
-
Entries recorded
Granulation
-
Mixing time recorded
-
Binder addition recorded
-
Granule consistency checked
Drying
-
Temperature recorded
-
LOD tested
-
Within specification
Blending
-
Lubricant added last
-
Mixing time recorded
-
Blend uniformity passed
Compression
-
Tooling inspected
-
Weight variation within limit
-
Hardness within limit
-
Friability < 1%
-
Disintegration within limit
Post-Operation
-
Reconciliation completed
-
Cleaning performed
-
BMR completed
Excel-Format Checklist Template (Column Structure)
You can directly paste this into Excel:
| Section | Activity | Standard/Limit | Checked (Yes/No) | Remarks | Sign | Date |
|---|---|---|---|---|---|---|
| Dispensing | Material verified | As per MBR | ||||
| Granulation | Mixing time | As per BMR | ||||
| Drying | LOD | NMT ___% | ||||
| Blending | Uniformity | RSD ≤ ___% | ||||
| Compression | Weight variation | ±5% | ||||
| Compression | Hardness | ___ kp | ||||
| Compression | Friability | NMT 1% | ||||
| Compression | Disintegration | ≤ ___ min | ||||
| Packing | Leak test | Pass |
Rotary Tablet Compression Machine Specific Checklist
Machine Setup
-
Punch & die inspected
-
Turret cleaned
-
Feeder alignment checked
-
Pre-compression force set
-
Main compression force set
-
Fill cam adjusted
-
Weight control calibrated
During Operation
-
RPM recorded
-
Output per hour monitored
-
Tablet weight every 15–30 min
-
Hardness monitored
-
Thickness monitored
-
Noise/vibration abnormality check
-
Lubrication system functioning
Troubleshooting Check
-
Capping observed?
-
Lamination?
-
Sticking/picking?
-
Double impression?
Post-Run
-
Tooling removed safely
-
Cleaning completed
-
Logbook updated
-
Reconciliation done
QA Audit Checklist for Regulatory Inspection
Documentation Review
-
MBR approved & controlled
-
BMR complete & reviewed
-
Deviations closed
-
CAPA implemented
-
Change control documented
Validation
-
Process validation report
-
Cleaning validation report
-
Equipment qualification
-
Analytical method validation
Data Integrity
-
No overwriting
-
Corrections signed & dated
-
Audit trail review performed
-
Electronic system validated
Quality Systems
-
Complaint handling system
-
Recall procedure
-
Supplier qualification
-
Stability study program
Regulatory Compliance
-
cGMP compliance (WHO/FDA/EU)
-
Batch traceability
-
Training matrix available
Key Features of the Tablet Processing Checklist
-
Raw Material Dispensing
-
Verification of material identity, quantity, and batch number
-
Compliance with approved Master Batch Record (MBR)
-
Weighing and documentation procedures
-
-
Sifting, Milling, and Granulation
-
Mesh size and sieve integrity checks
-
Wet and dry granulation process monitoring
-
Moisture content and granule consistency checks
-
-
Drying
-
Temperature and duration monitoring
-
Loss on drying (LOD) verification
-
Uniformity checks
-
-
Blending and Lubrication
-
Blend uniformity assessment
-
Correct sequence and timing for lubricant addition
-
-
Compression
-
Tablet press setup and tooling inspection
-
In-process checks: weight, thickness, hardness, friability, disintegration
-
Monitoring for capping, lamination, sticking, or picking
-
-
Coating (If Applicable)
-
Coating solution preparation
-
Pan speed, spray rate, and temperature control
-
Weight gain monitoring and visual inspection
-
-
Inspection and Packing
-
Visual and mechanical inspection
-
Metal detection
-
Packaging material verification and batch coding
-
-
Cleaning and Documentation
-
Post-process cleaning and line clearance
-
Complete record-keeping for regulatory compliance
-

















